Principles of green chemistry
The Bhopal disaster
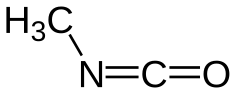
Methyl isocyanate (MIC) is a highly toxic, volatile, and flammable organic compound (CH3NCO) used primarily as an intermediate in the production of carbamate pesticides, polyurethane foam, and plastics. It is notoriously hazardous, causing severe respiratory, eye, and skin irritation
What is green chemistry?
Using renewable resources
Using renewable resources is the first key point in green chemistry.
Ethanol, C2H5OH, is often used as a solvent for chemical reactions. It is also a raw material in the synthesis of useful compounds such as pharmaceuticals and pesticides, and can be used as a fuel.
Ethanol can be made by the reaction of ethene (derived from crude oil) and water. Alternatively, it can be made by fermentation of biomass.

Exam Question #1
Alternatives to hazardous chemicals
Many reactions in the chemical industry use a solvent to dissolve the raw materials
Some solvents can be flammable and toxic, so safer alternatives are being researched.
An example of a safer solvent is the use of liquid carbon dioxide in dry cleaning. This is an alternative to the solvent 1,1,2,2-tetrachloroethane, which is toxic and harmful.
The Chemistry of Solvents
Atom economy
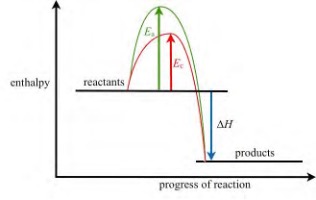
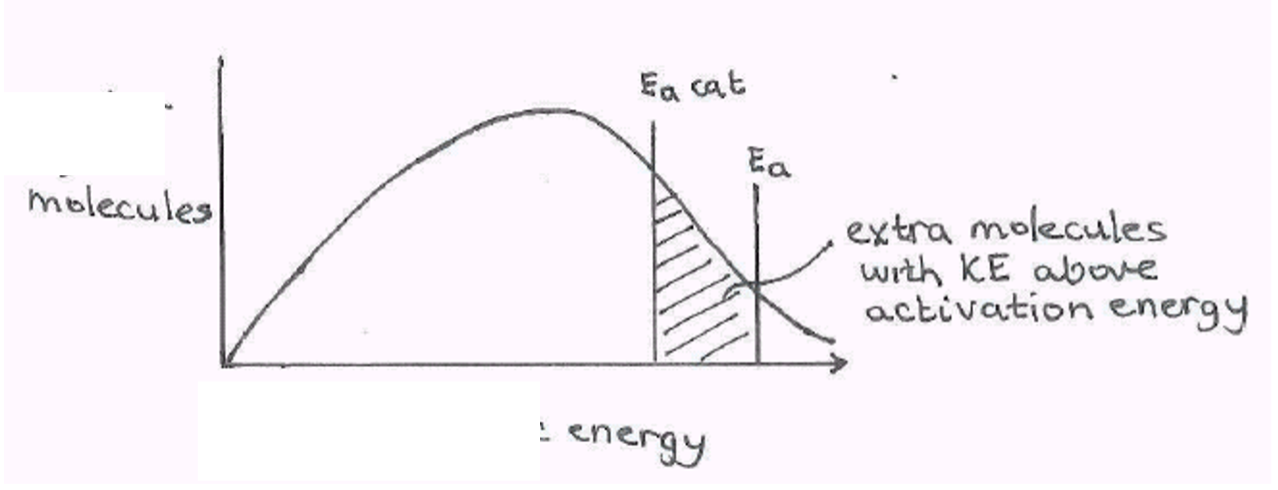
Use catalysts when possible
Common industrial catalysts include iron, platinum, vanadium(V) oxide and nickel. Enzymes can also be used as catalysts in industrial processes.
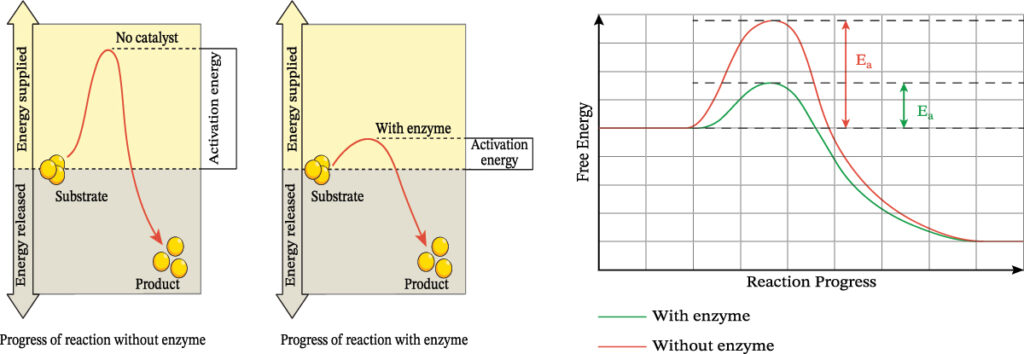
Catalysts act by providing an alternative route for a reaction with a lower activation energy. At any given temperature, more particles are likely to have enough energy to react when they collide.
Only a small amount of a catalyst is needed in a reaction, and it can be recycled and reused multiple times.
Enzyme or metal?
Exam Question #2
Exam Question #3
Energy is used in the chemical industry for heating, separating and drying.
Heat is often produced by burning fossil fuels. However, some reactions can now be carried out using microwaves to generate the heat needed.
There is also the potential for using solar power for heating and for water purification.

What are the advantages and disadvantages of using microwaves or solar power for heating?
The advantage of solar panels is that they use a renewable source of energy. The disadvantages are that enough space is needed to install them and the Sun doesn’t always shine. Because they are so large, it is only worthwhile to install them if they produce enough power for large scale use.
Reducing water use

Reducing pollution

Reduce and recycle waste

Which is greener?
Greenhouse gases
Global warming: true or false?
What are greenhouse gases?
Global warming
Global warming potential
Not all greenhouse gases contribute to global warming to the same degree.
The global warming potential (GWP) is a measure of how much a given gas can contribute to global warming over a given time period. It is measured relative to carbon dioxide, which is given a global warming potential of 1.
The GWP of a gas is calculated using the following factors:
Greenhouse gases compared
The contribution of a greenhouse gas to global warming depends on its global warming potential (GWP) and the amount of that gas present in the atmosphere.
|
Greenhouse |
Average % of |
After |
After |
|---|---|---|---|
|
water vapour* |
1–4 |
– |
– |
|
carbon dioxide |
0.038 |
1 |
1 |
|
methane |
0.0002 |
72 |
25 |
|
nitrogen |
0.00003 |
289 |
298 |
*Water vapour is estimated to be the most powerful of these greenhouse gases. Its concentration in the atmosphere varies depending on the temperature so no GDP is calculated for it.
The local effects of climate change
Exam Question #4
Causes of climate change
Causes of climate change – class activity
How scientists can help
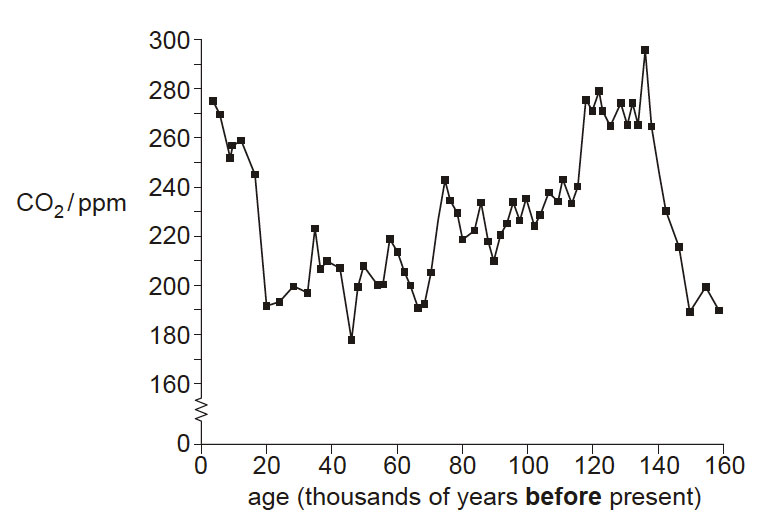
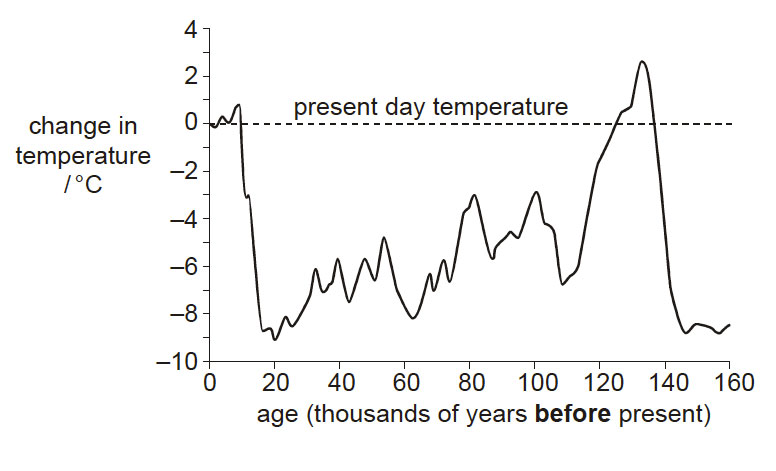
It is now widely accepted that human activity is causing an increase in the concentration of carbon dioxide in the atmosphere, and that this is leading to global warming.
What is the role of scientists in combating global warming?
What do you think? – class activity
Carbon footprints
What are carbon footprints?
Carbon footprint is a measure of the impact of a person’s activities on the environment, in terms of how much CO2 is produced by each activity.
|
Country |
Average carbon footprint radius |
|---|---|
|
USA |
13 – 15 |
|
Australia |
14 – 21 |
|
UK |
9 – 11 |
|
China |
7 – 9 |
|
India |
1.2 – 2 |
Carbon footprints
Reducing your carbon footprint
What is carbon neutral?
Carbon neutral describes any activity that produces no overall emissions of CO2.
There are two ways of working towards carbon neutrality:
Carbon offsetting is the practice of balancing activities that emit CO2 with activities that absorb it; for example, planting trees that take in CO2 as they grow.
Carbon offsetting is controversial because the act of planting trees in large numbers uses energy and emits CO2 even if the trees themselves absorb it.
Coldplay’s “Music of the Spheres” world tour was designed to be their most sustainable yet by focusing on three core pillars:
Are biofuels carbon neutral?
Advantages of biofuels
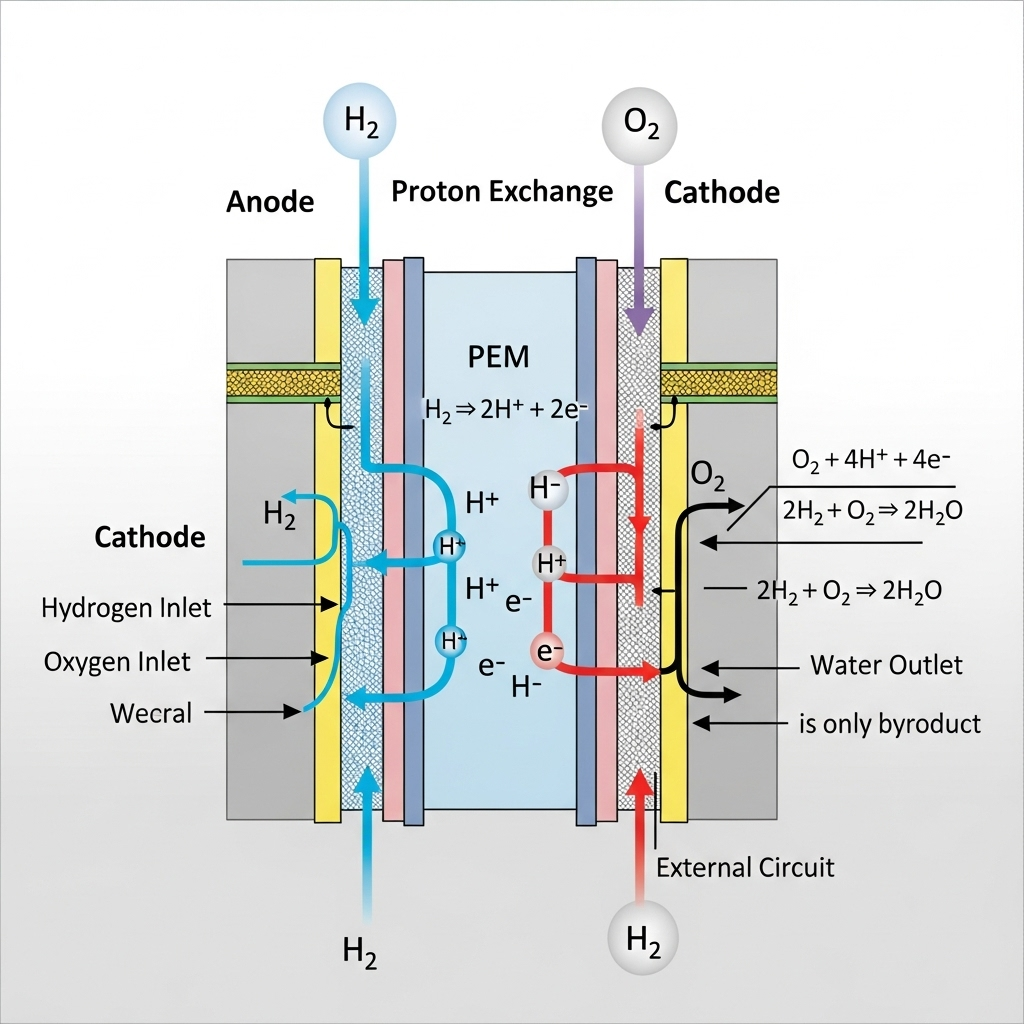
Is hydrogen carbon neutral?
When hydrogen is burned as a fuel, the only product is water.
Fuel cells that use this method of energy production can be incorporated into engines, as shown on the right.
The hydrogen fuel is generated by the electrolysis of water.
Do you think hydrogen is a carbon neutral fuel?
Hydrogen is generated for fuel by the electrolysis of water. This uses electricity, which must be generated somehow, and most often by the burning of fossil fuels. Although hydrogen is, therefore, cleaner than fossil fuels, it is not carbon neutral. Students should be able to reach this decision after some discussion.
CFCs and the ozone layer
What are CFCs?
Chlorofluorocarbons (CFCs), are a family of organic compounds.
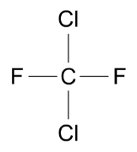
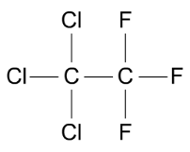
In the 1930s, CFCs were developed for use as refrigerants, to cool the air inside refrigerators.
They were then found to be useful in many other products, including aerosol sprays and foam fire extinguishers.
What is the ozone layer?
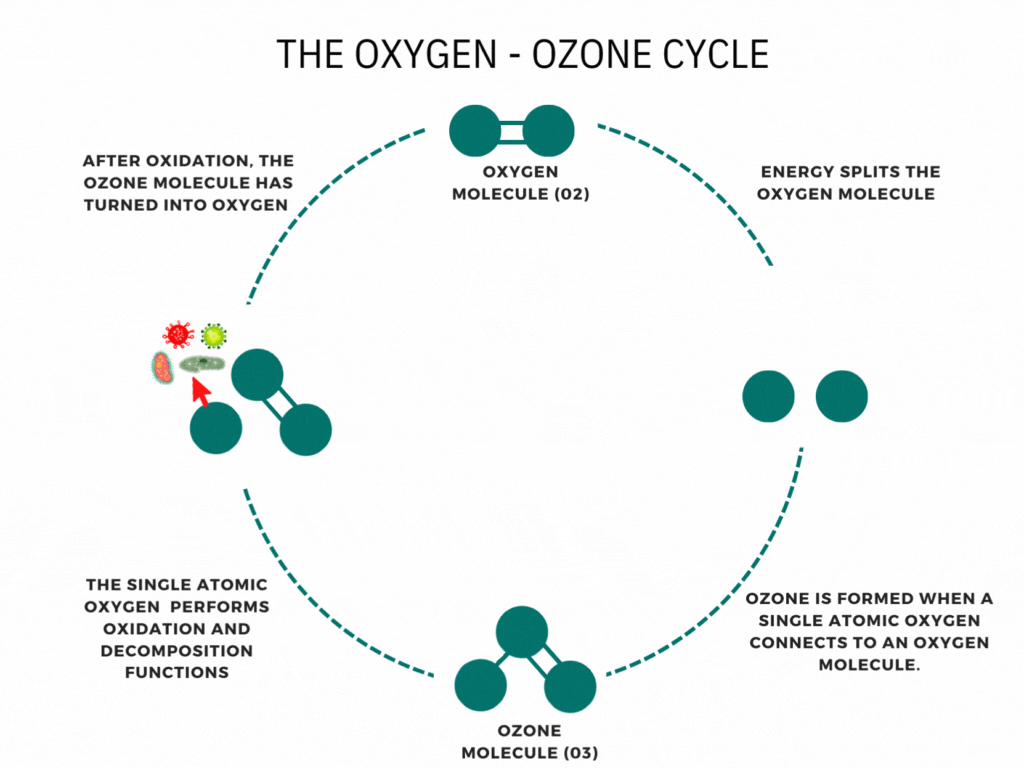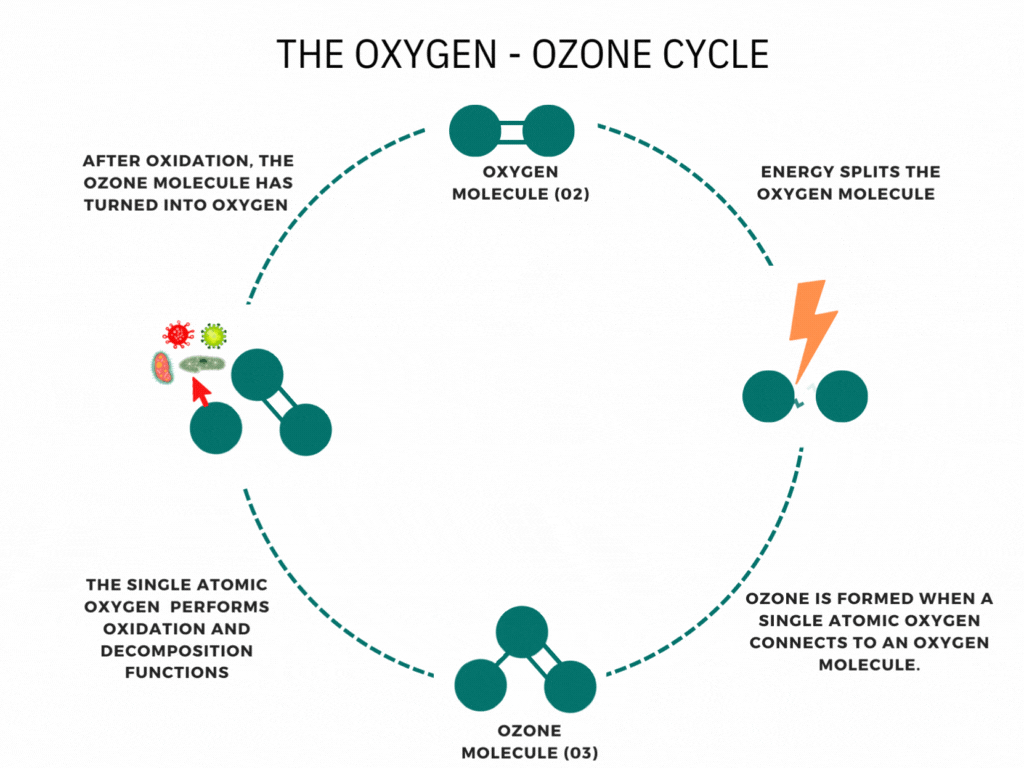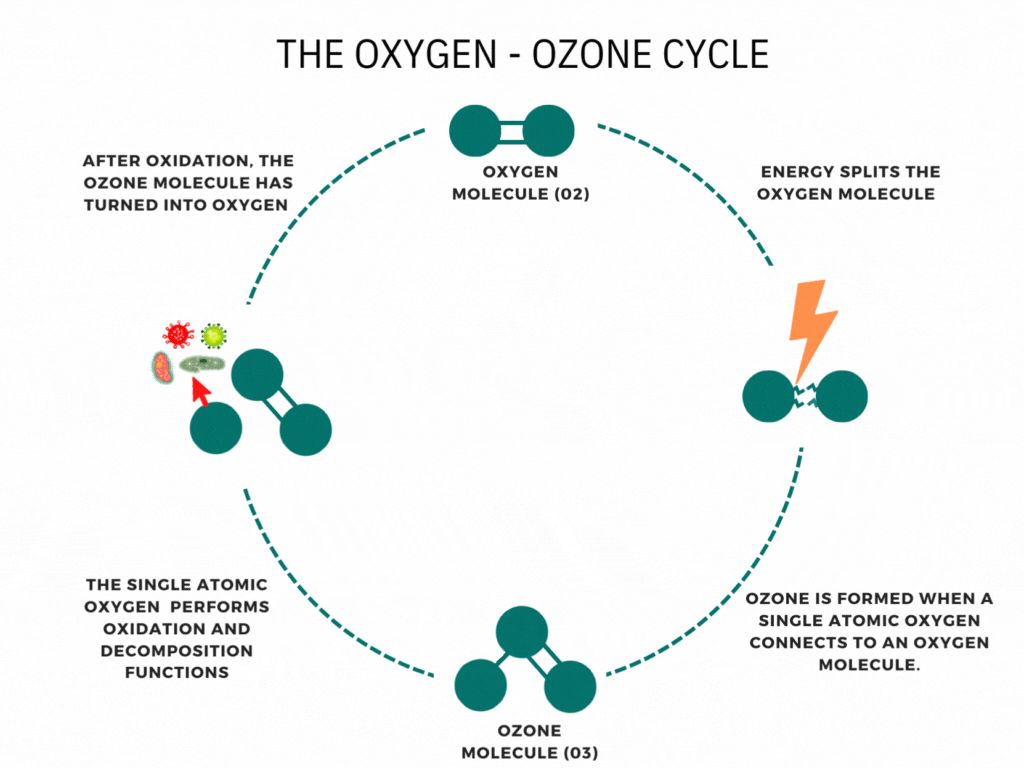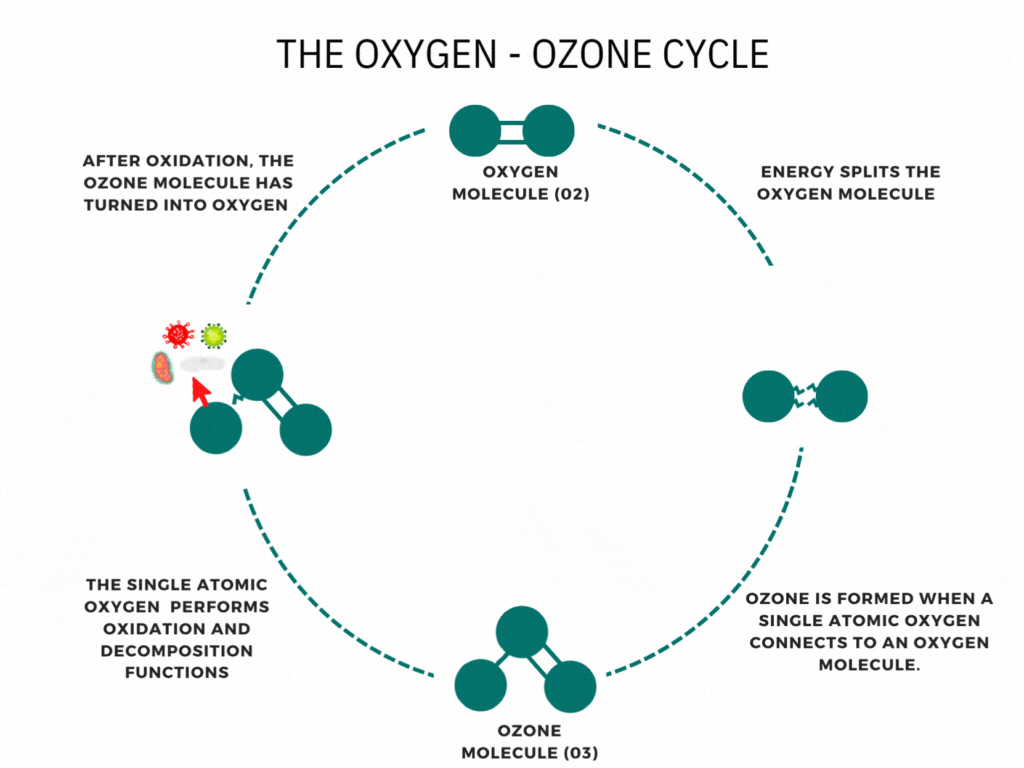
CFCs and ozone
At ground level, CFCs are very stable. This is what allows them to exist in the atmosphere for long enough to reach the stratosphere. Once there, they absorb UV radiation from the Sun, which breaks their covalent bonds to form highly reactive free radicals.
CCl2F2 → •CClF2 + Cl•
These react very quickly with ozone molecules in a free radical chain reaction:
Cl• + O3 → ClO• + O2
ClO• + O3 → Cl• + 2O2