The atmosphere
Introduction
Pollution Types & Sources
How do human activities affect the air we breathe?
Why is the atmosphere important?
The Earth is different to the other planets in our solar system because it has an atmosphere that can support life.
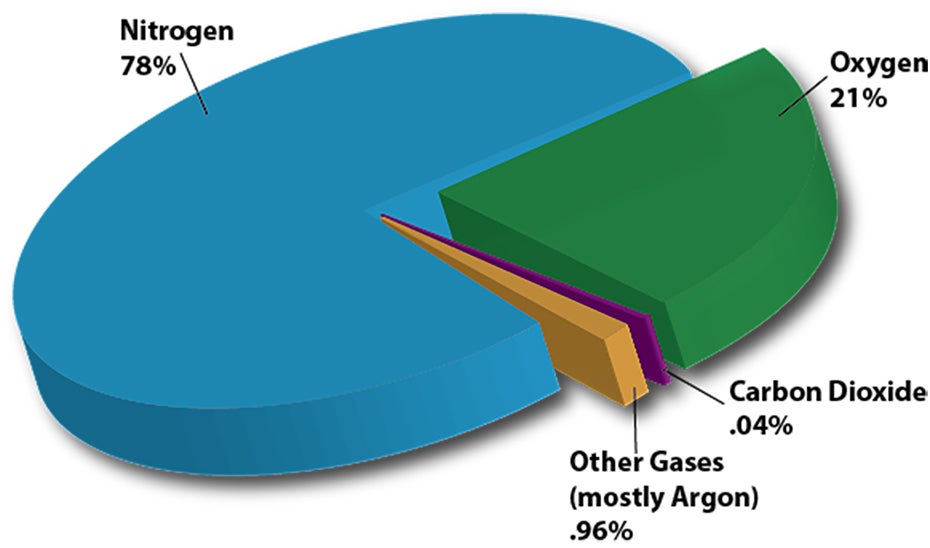
The atmosphere is an envelope of different gases (air) surrounding Earth.
80% of atmospheric gases are in the 15 km closest to Earth. This is a very thin layer compared to the Earth’s diameter, which is 12,756 kilometres.
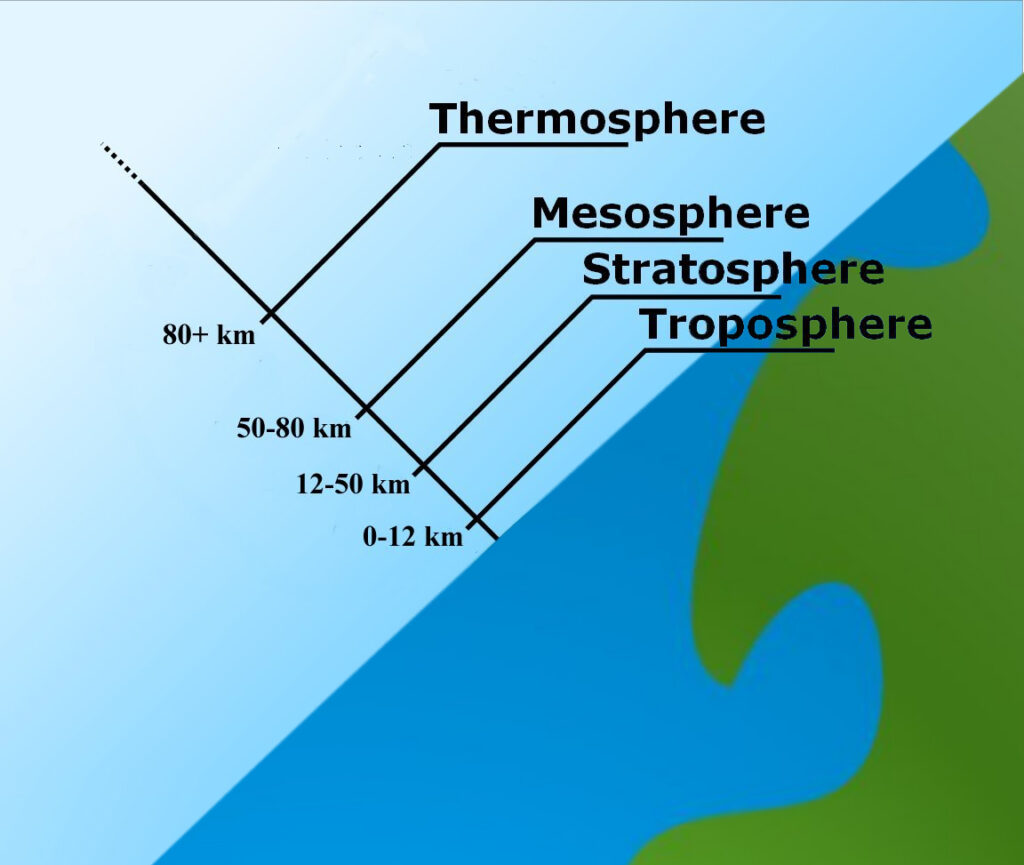
The Troposphere
The troposphere is the lowest layer of Earth’s atmosphere. Most of the mass (about 75-80%) of the atmosphere is in the troposphere. Most types of clouds are found in the troposphere, and almost all weather occurs within this layer. The troposphere is by far the wettest layer of the atmosphere (all of the other layers contain very little moisture).
Exam Question #1
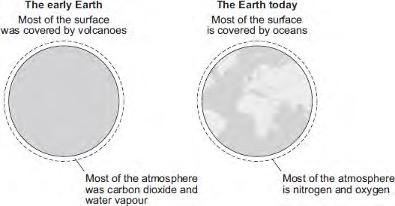
The history of the atmosphere
Evolving atmosphere
Exam Question #2
Air Pollution
What is air pollution?
Air pollution is the presence of substances in the air that are harmful to health or the environment.
It can be easy to spot air pollution in cities, but its effects are not limited to urban areas because air circulates freely all over the world.
Air pollution can cause the destruction of forests, death of fish in lakes and premature death in humans.
How does air pollution cause so many problems?
What causes air pollution?
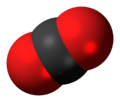
Carbon dioxide
Carbon dioxide is a chemical compound with the chemical formula CO2. It is made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms.
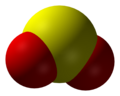
Sulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula SO2. It is a colorless gas with a pungent smell that is responsible for the odor of burnt matches.

Carbon particulates
carbon particulates is the light-absorbing refractory form of elemental carbon remaining after pyrolysis (e.g., charcoal) or produced by incomplete combustion (e.g.soot).

Nitrogen monoxide
Nitric oxide (nitrogen oxide, nitrogen monooxide, or nitrogen monoxide) is a colourless gas with the formula NO. It is one of the principal oxides of nitrogen.

Carbon monoxide
Carbon monoxide (chemical formula CO) is a poisonous, flammable gas that is colorless, odorless, tasteless, and slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond.
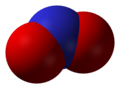
Nitrogen dioxide
Nitrogen dioxide is a chemical compound with the formula NO2. One of several nitrogen oxides, nitrogen dioxide is a reddish-brown gas.
Not to be confused with nitric oxide (formula NO), nitrous oxide (N2O), or generic nitrogen oxide pollutants NOx.
Causes and effects of air pollutants
How is carbon dioxide produced?
The element carbon is found in all living things and moves through the environment in a chain of reactions known as the carbon cycle.
In this cycle, the chemical reactions that take place during respiration and burning fossil fuels produce carbon dioxide.
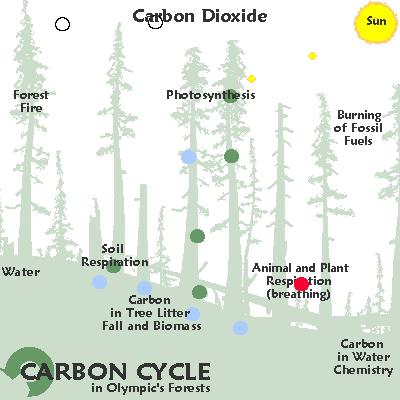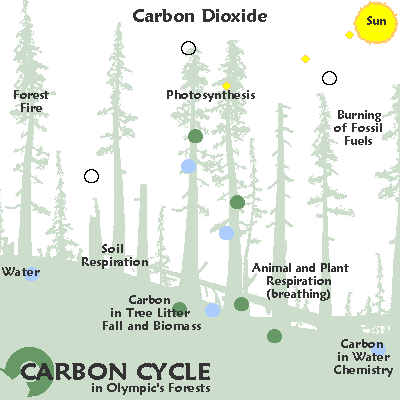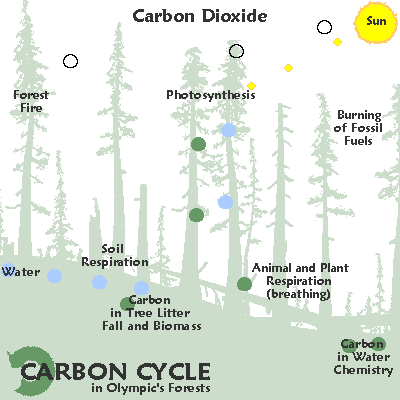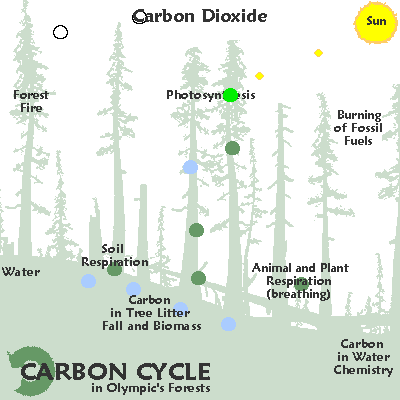
Carbon dioxide is an important atmospheric gas as it prevents heat radiation produced by the Earth from escaping into space.
This is the greenhouse effect, which makes the Earth warm enough for life.
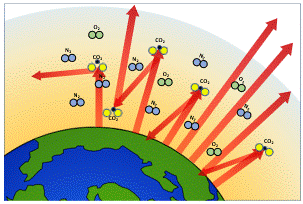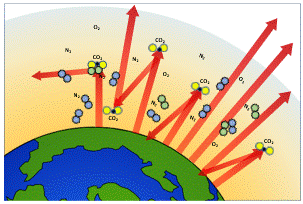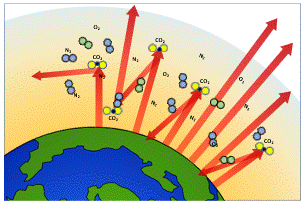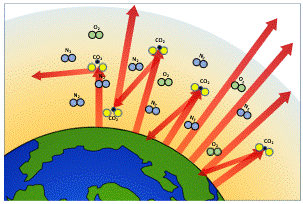
However, increased levels of carbon dioxide in the atmosphere could cause dramatic changes to the Earth’s climate.
How do fossil fuels cause air pollution?
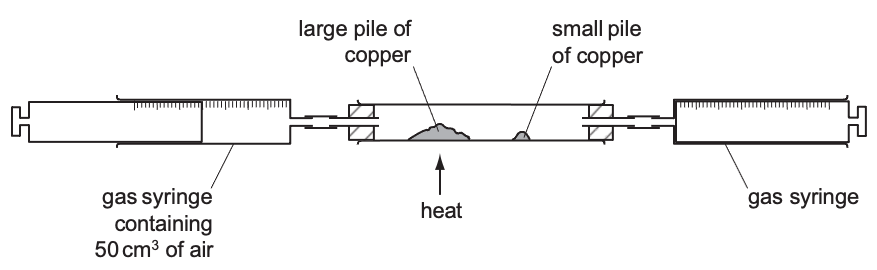
When fossil fuels are burnt fully in oxygen, the chemical reaction that occurs is called complete combustion.
fossil fuels + oxygen → carbon dioxide + water
The problems with burning fossil fuels include:
Exam Question #3
What is incomplete combustion?
If there is not enough oxygen present when fossil fuels are burned, incomplete combustion occurs.
This reaction produces carbon and carbon monoxide gas.
Carbon monoxide gas is extremely harmful to human health because it stops blood from carrying oxygen.
What is acid rain?
Acid rain is rain, or precipitation, with a pH of 5.6 or lower. It is caused by the pollutants sulfur dioxide and nitric oxides.
The term ‘acid rain’ was first used by the Scottish scientist, Robert Angus Smith, in the 19th century.
He conducted air quality research in Manchester, one of the first industrialized cities in the world.
Acid rain causes millions of pounds worth of damage to buildings in the UK each year.
It is also harmful to the environment and can destroy forest habitats.
Causes and effects of acid rain
Exam Question #4
Summary of air pollutants
What are the causes and effects of these air pollutants?
|
Pollutant |
Cause |
Effect |
|---|---|---|
|
carbon dioxide |
combustion and respiration |
adds to the greenhouse effect |
|
nitric oxides |
high temperature combustion |
acid rain and smog |
|
carbon monoxide |
incomplete combustion |
lowers blood’s ability to carry oxygen |
|
sulfur dioxide |
burning coal |
acid rain |
|
particulates |
burning wood and fossil fuels |
deposits as soot and lung damage |
Health issues
How does air pollution affect health?
Weather reports often include warnings about high air pollution levels. Why is this?
Normally, pollution levels in the UK are not high enough to cause problems to people’s health.
However, high pollution levels have been shown to irritate existing heart and respiratory problems. Air pollution can even cause premature death in the vulnerable or elderly.
People who know they are sensitive to pollutants are advised to take precautions when air pollution levels are high, such as not to exercise outside.
What is smog?
Smog is a mixture of air pollutants and particulates that is sometimes found in the lower levels of the atmosphere.
It can reach dangerous levels in built-up areas.
Smog is more common in summer because hot weather acts as a catalyst in the formation of smog components.
A large part of smog is ground level ozone (O3). This is a highly toxic gas that can damage the lungs.
Ozone is formed when nitrogen oxides and volatile organic compounds combine with oxygen.
Reducing pollution
Why should we control pollution?
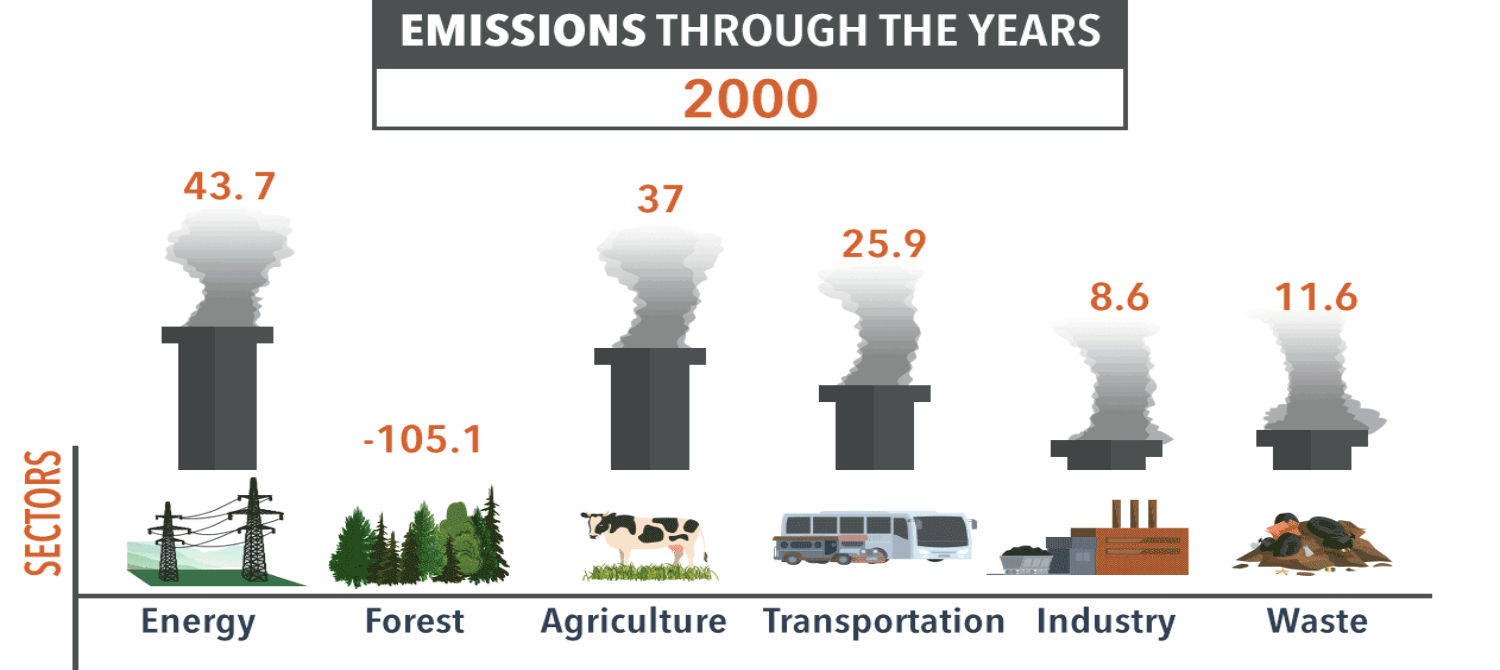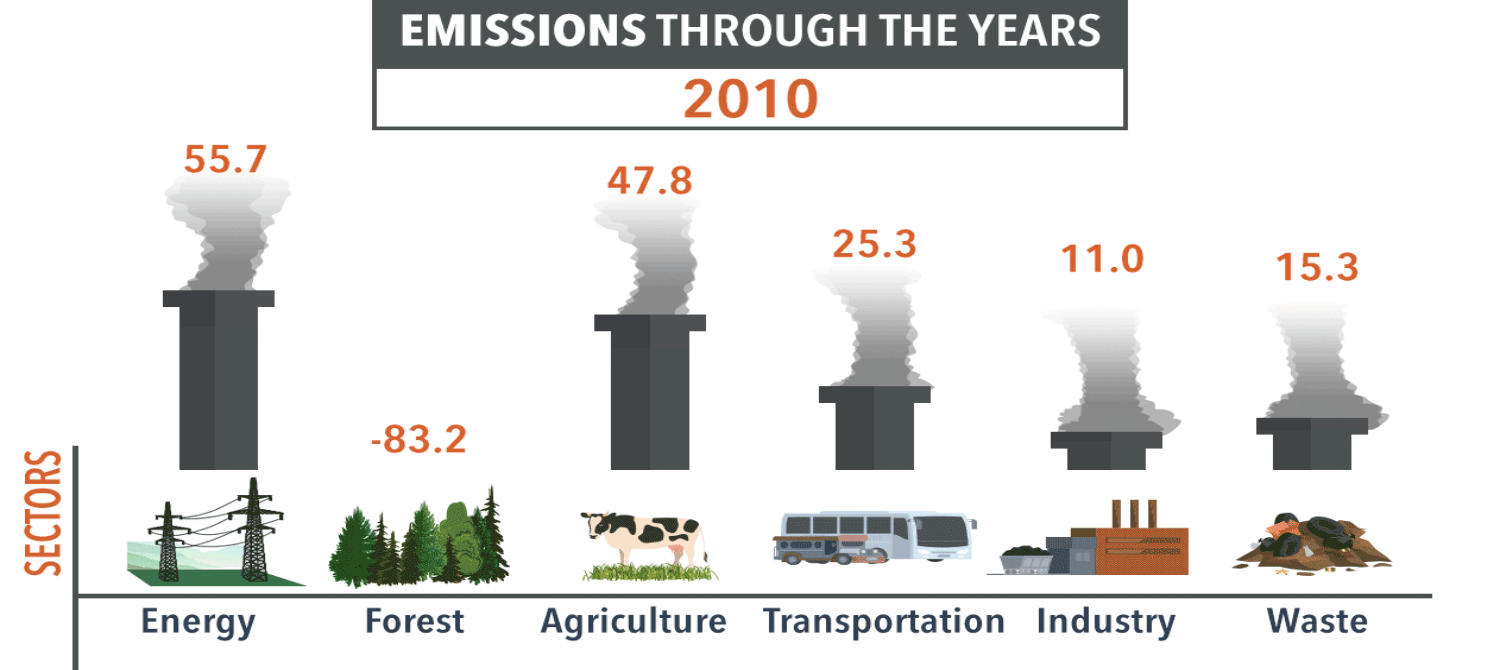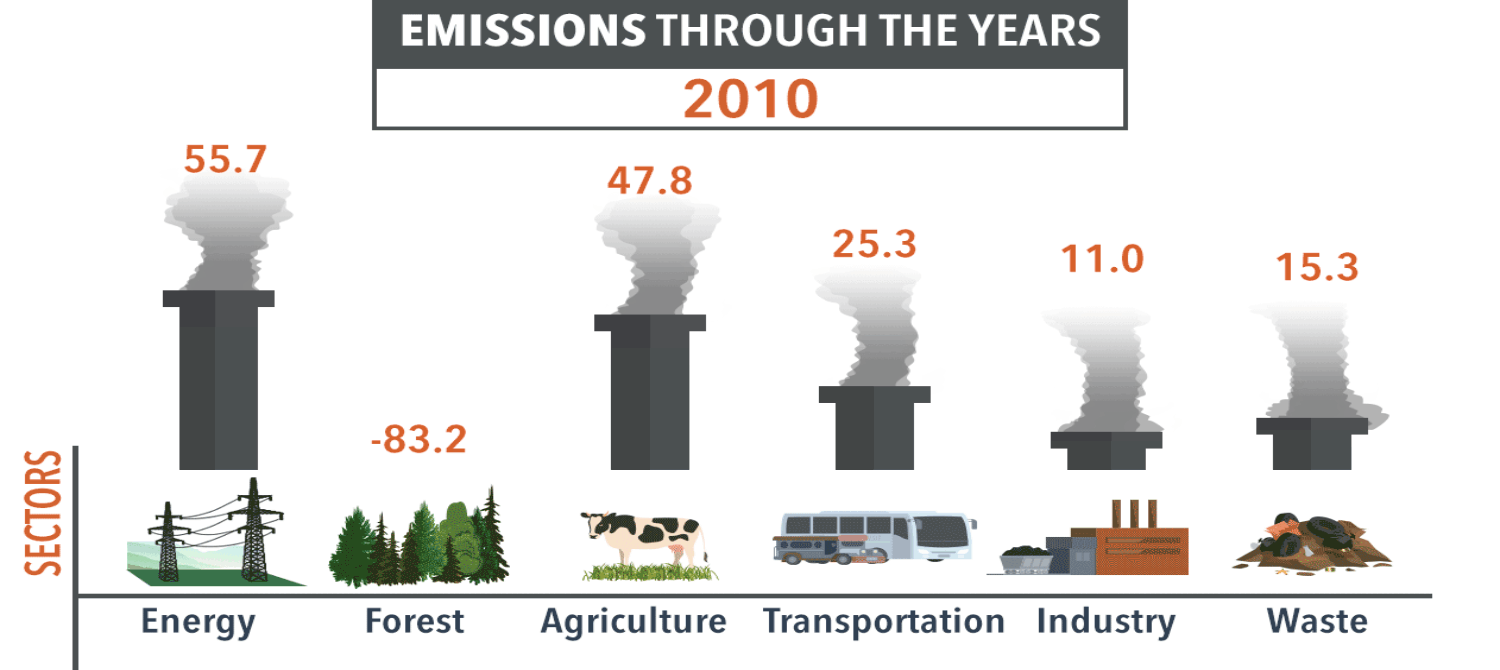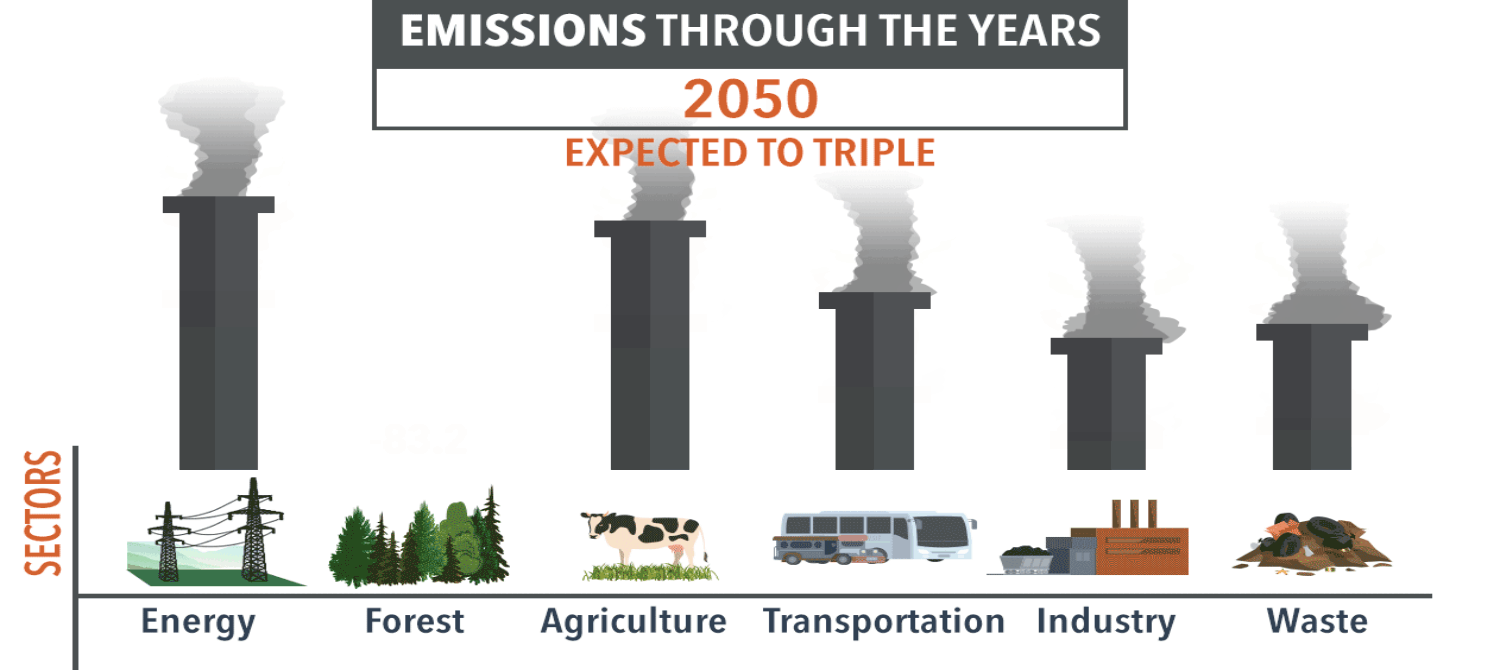
Human actions can cause or reduce air pollution, which is not just a current problem.
For example, severe smog in London caused the premature death of 12,000 people in 1952.
Most of the smog was formed by high levels of particulates in the air, caused by burning wood and coal for heating.
The Clean Air Act of 1956 created smoke-free zones in cities.
Power plants were moved to rural areas, reducing city smog. How else have human actions improved the atmosphere?
Is there a hole in the ozone layer?
The ozone layer in the upper atmosphere filters UV radiation.
In the 1980s, scientists discovered that ozone was being destroyed by the chlorofluorocarbons (CFCs) widely used in aerosols and refrigerators. CFCs can stay in the environment for 50 years, destroying ozone long after they are produced.
However, international regulations to reduce CFC emissions may be helping to repair the ozone layer.
The ozone layer continues to heal, with projections indicating a return to 1980 levels by approximately 2066 over Antarctica and 2040 for the rest of the world, following a trend of smaller, shorter-lived holes in 2024 and 2025. Global monitoring, including NASA Ozone Watch reports for early 2026, confirms that the phase-out of ozone-depleting substances is effective.
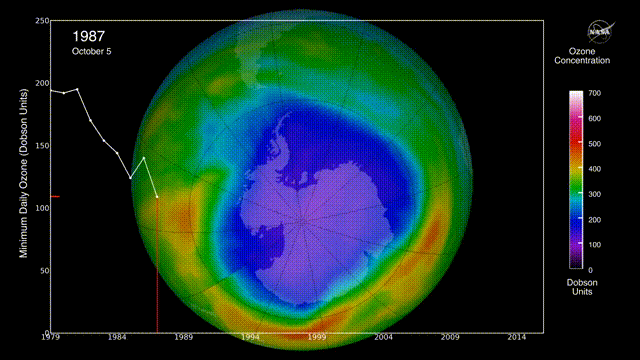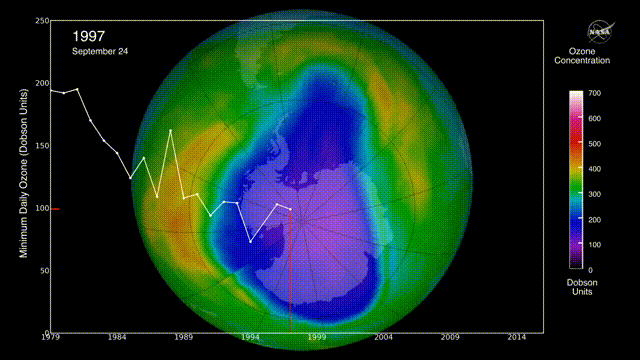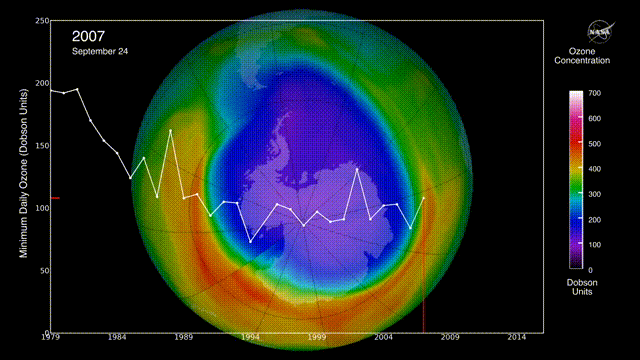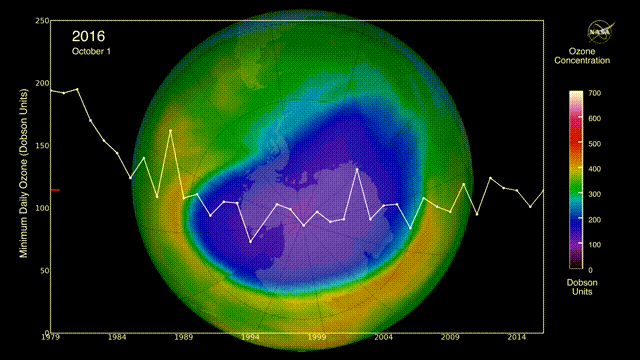
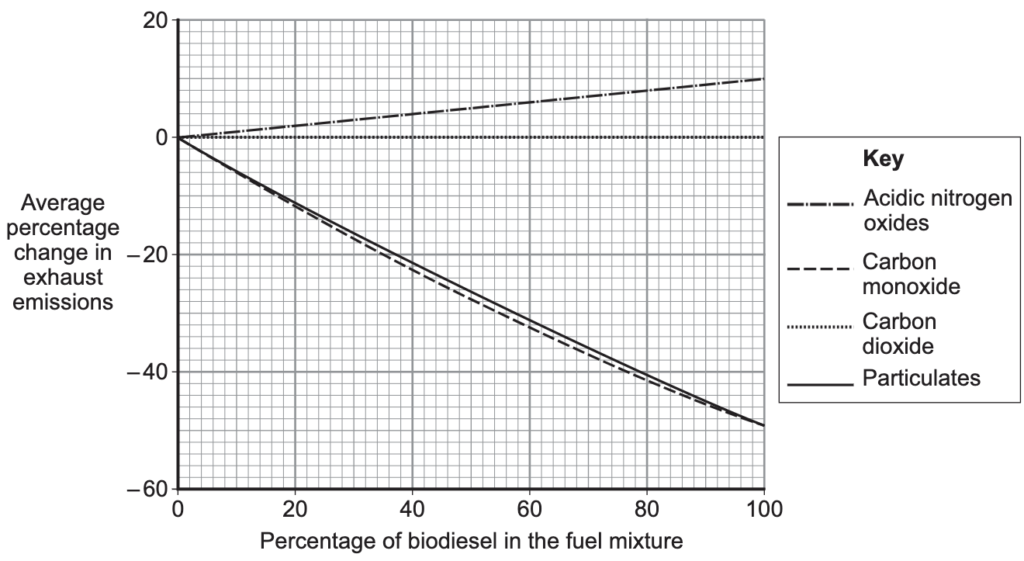
Can air pollution be reduced?
This summary provides a selection of the key information from the full publication, ‘Air quality statistics’. This publication contains statistics which help describe the trends in concentrations of air pollutants using a variety of metrics for the period 1987 to 2024.
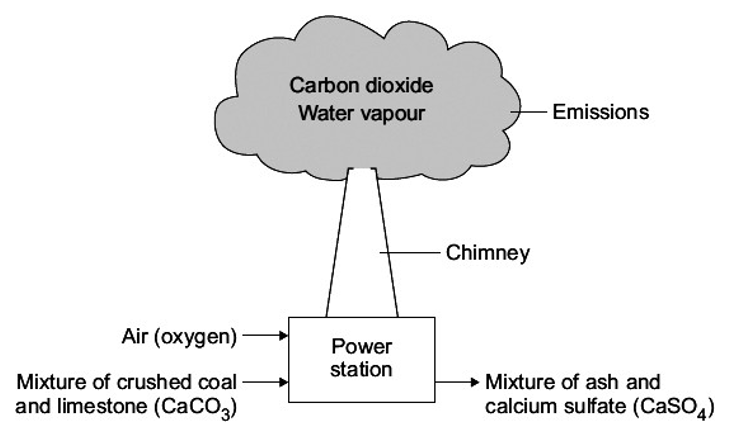
How can coal be made cleaner?
Sulfur dioxide emissions from coal can be reduced in two ways: by removing sulfur from the coal before combustion or by filtering the emissions produced by combustion.
A process called flue gas desulfurization is used to filter emissions.
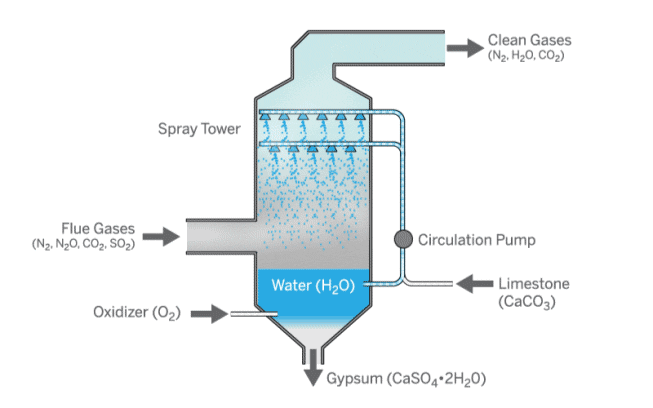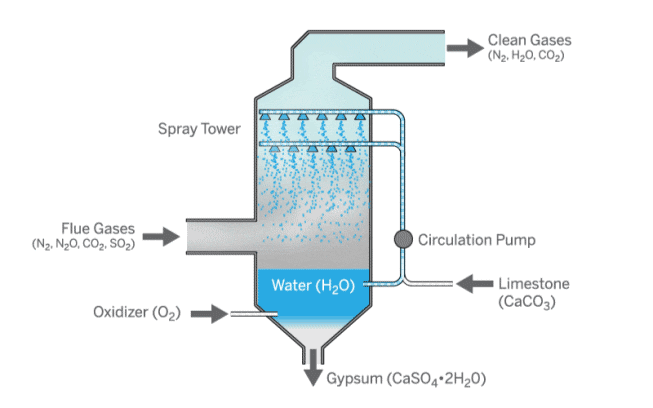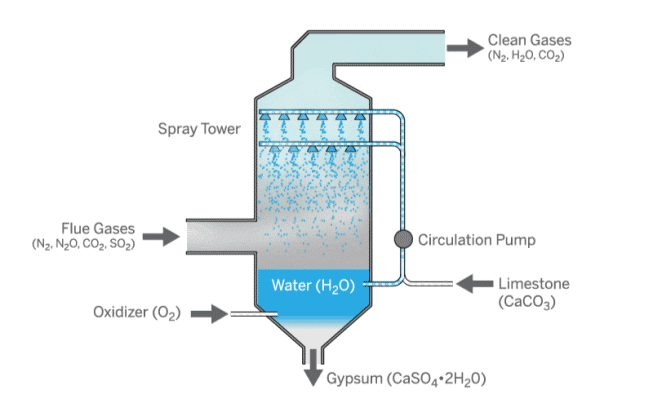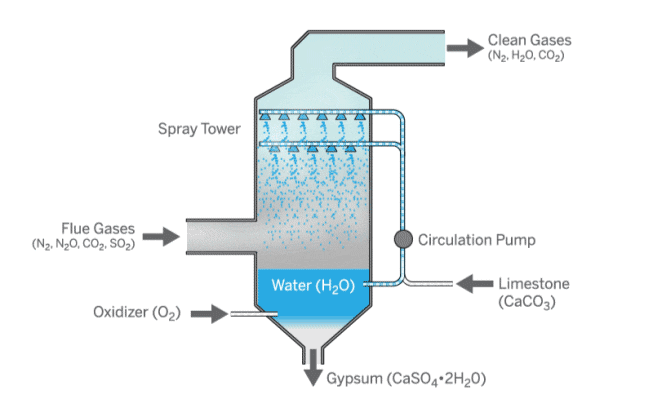
Sulfur dioxide in the flue gas is removed by reacting it with limestone and water.
This forms calcium sulfate, also known as gypsum, which can be used in construction.
How can vehicle pollution be reduced?
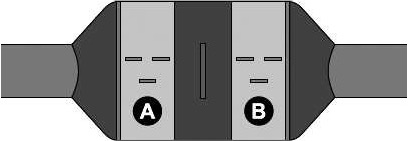
A catalytic converter is a device that is fitted to the exhaust pipe of a car to clean up exhaust fumes.
The inside of the converter has a honeycomb structure, covered in a metal catalyst, such as platinum or palladium.
This structure maximizes contact between the exhaust fumes and the metal catalyst to change the pollutants from combustion into less harmful substances.
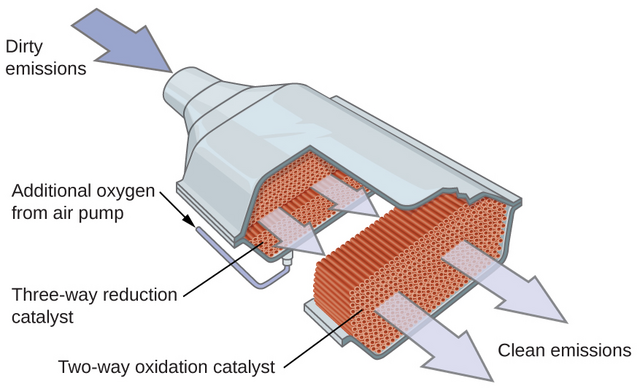
Word Equation: carbon monoxide + oxygen → carbon dioxide
Balanced Symbol Equation: 2CO + O2 → 2CO2


+

→